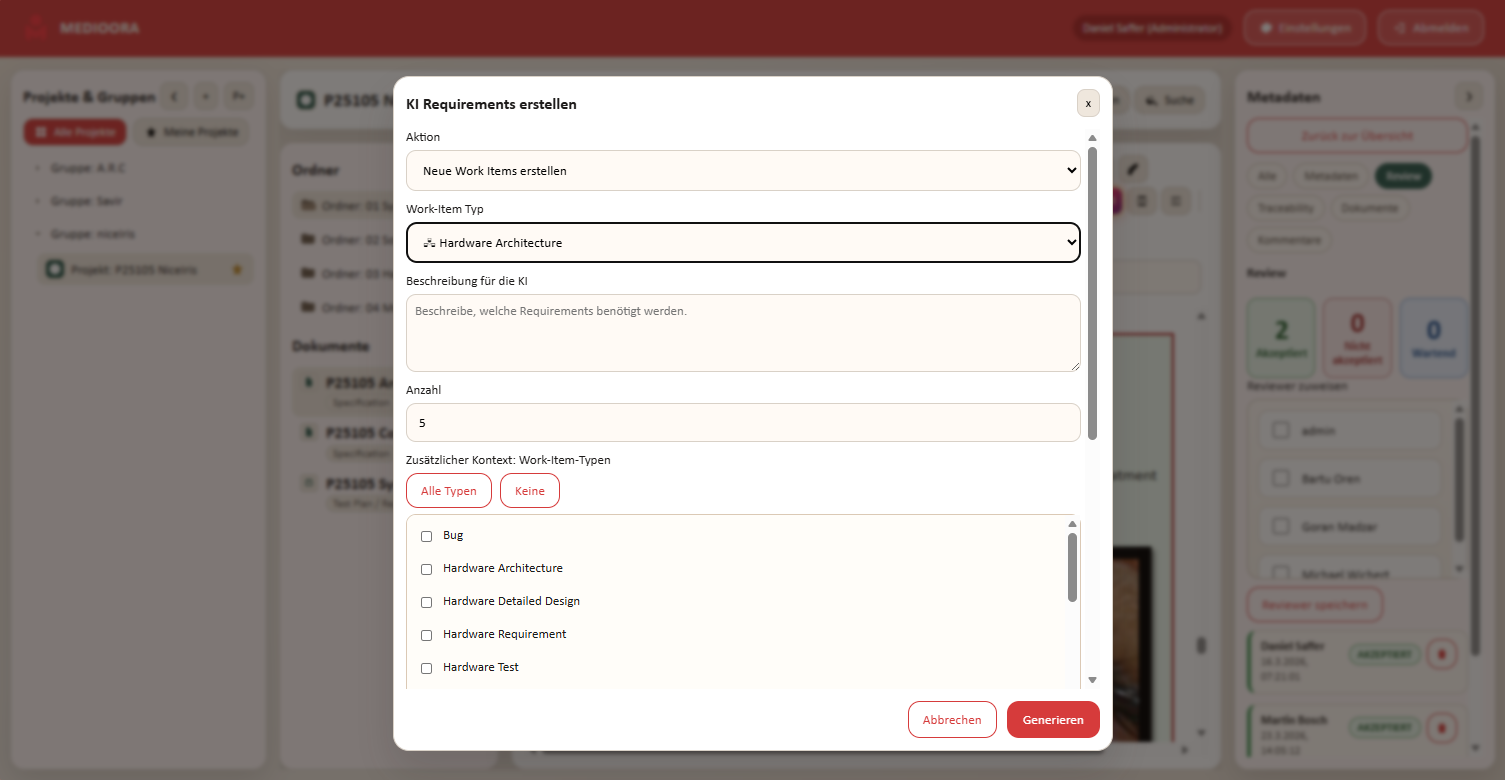
Das Requirements-Tool für sichere Medizintechnik
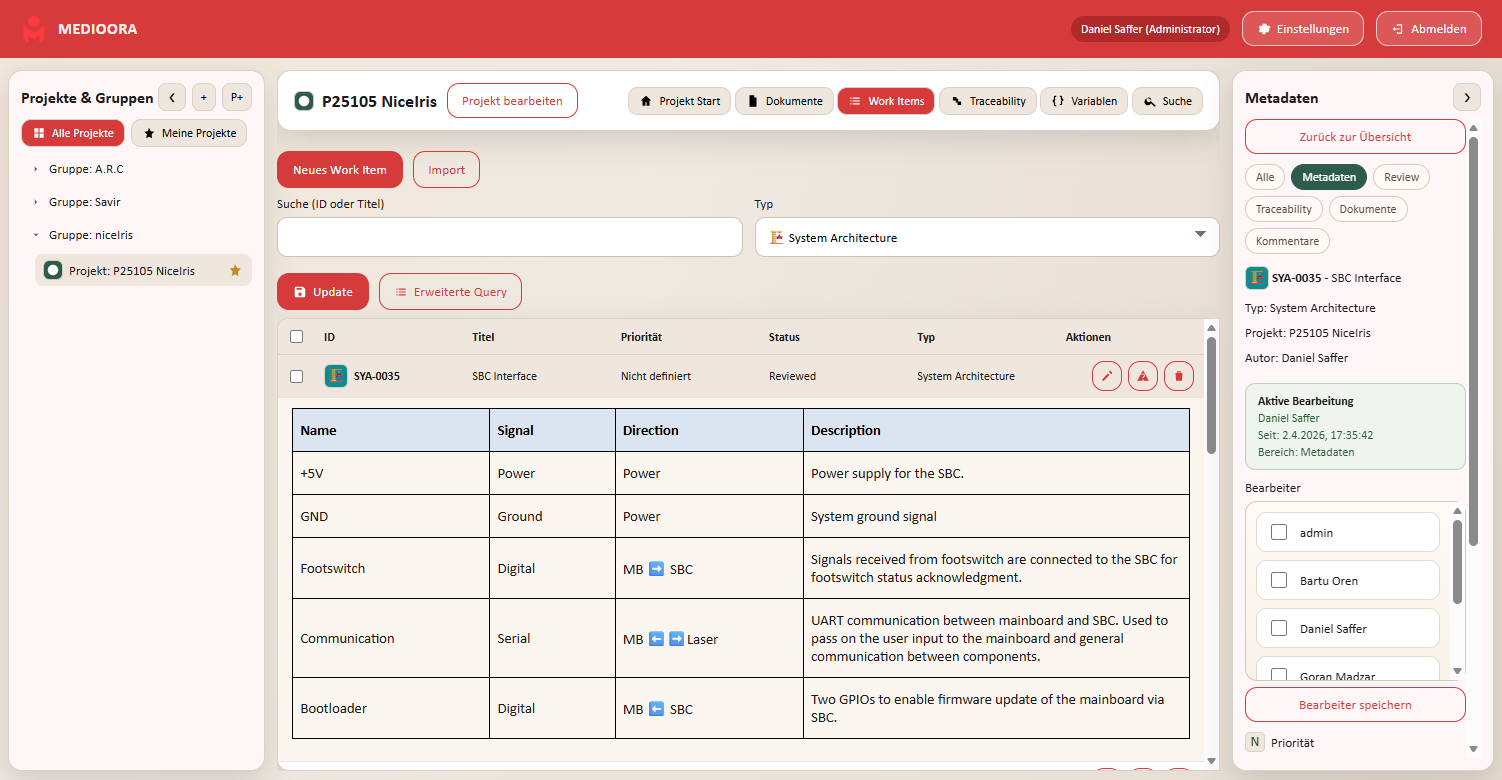
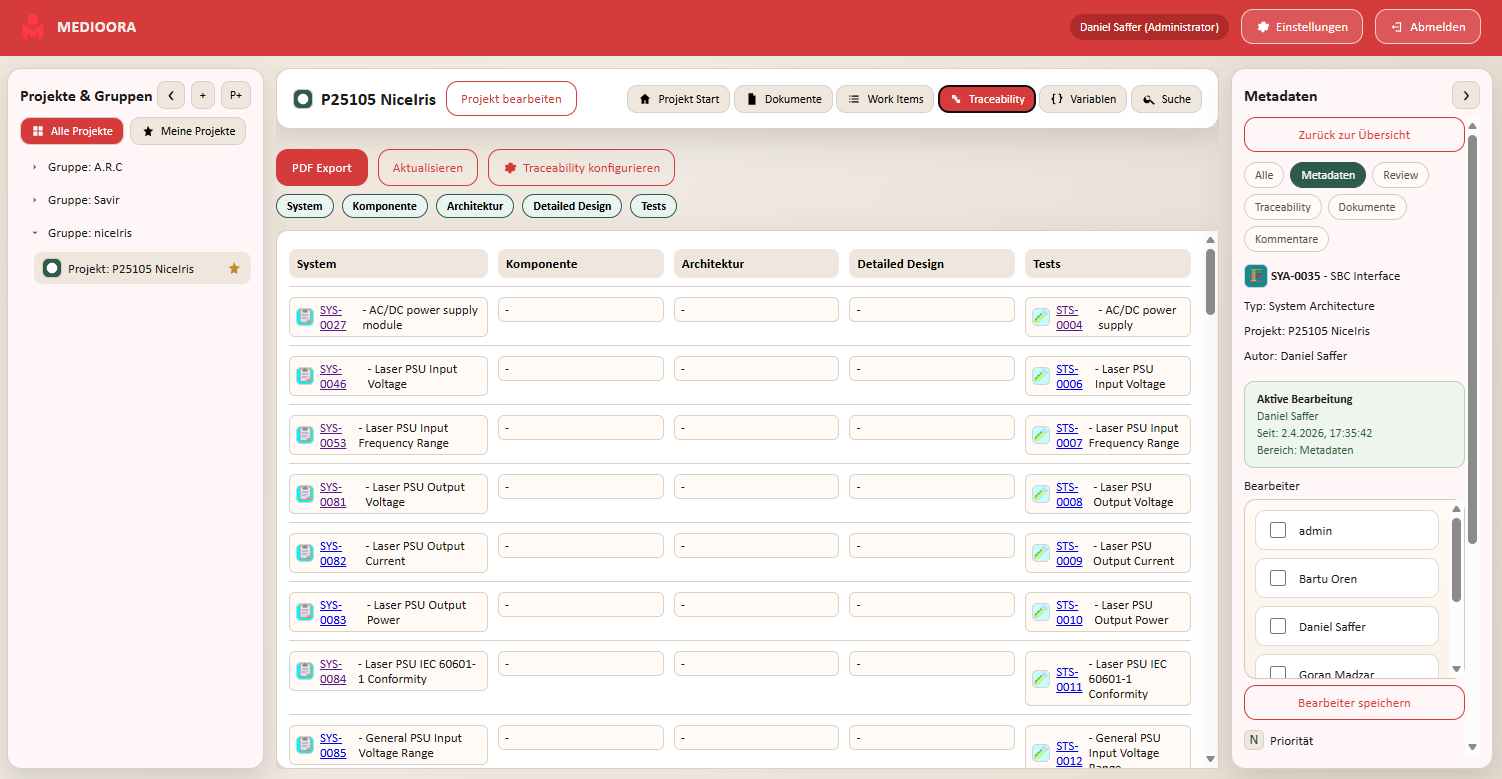
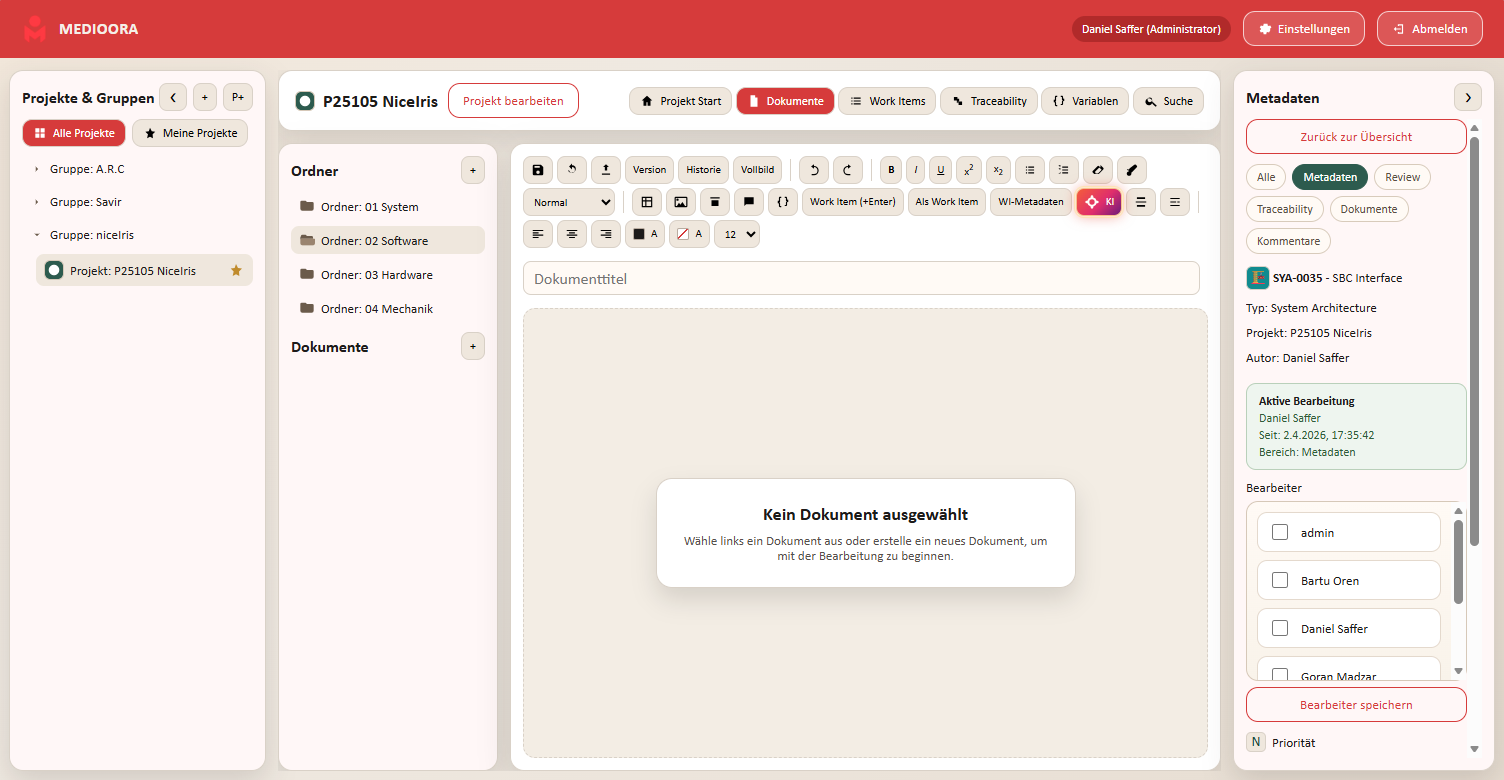
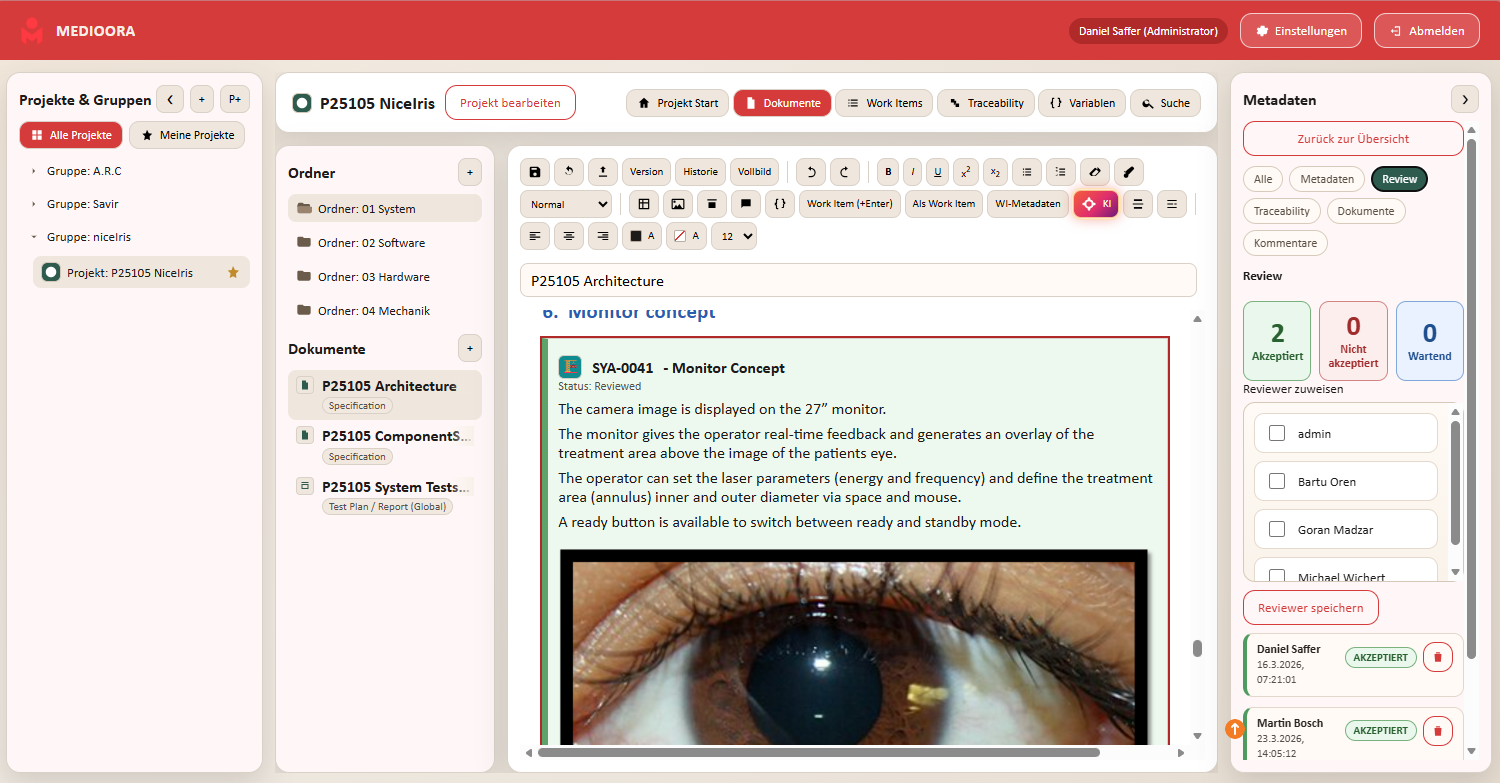
Medioora verbindet Requirements, Tracing, Design und Testing in einer zentralen Plattform. Für durchgängige Nachverfolgbarkeit, regulatorische Sicherheit und effizientere Entwicklung von Medizinprodukten.
- Persönliche Beratung für Ihr Projekt
Teams up and running in 3 weeks
Full setup package including configuration, onboarding and go-live support.
Workspace Setup
Setting up the workspace and importing existing templates.
Onboarding admins and users
Roles, permissions, processes and hands-on training for all teams.
Go-live, support and first requirements
Joint go-live, rapid support and the start of first requirements.
Frequently asked Questions & Answers
Quick answers to the most important questions about Medioora.
Medioora is for teams in regulated industries who want to manage requirements, development, testing and evidence in a structured way.
With our setup package, you’ll be up and running in three weeks: setup, onboarding and go-live with hands-on support.
Requirements management, tracing, tasks, collaboration, versioning, documentation and releases in one central platform.
Yes. Existing templates can be imported and saved as team standards in your workspace.
We guide admins and users hands-on through roles, permissions, workflows and daily use of the tool.
Security and traceability are at the core. Access, changes and approvals can be documented transparently.
Yes. We support you at launch, help prioritise initial requirements and assist with refining your processes.
No. The platform is designed for rapid adoption and is rolled out iteratively with your team.